Contract Research Service Center conducts animal experiments at a client’s request in accordance with quality assurance standards and delivers the results as a report. We have abundant background data for animal models which developed in CIEM, such as NOG mice as a severe immunodeficient model, rasH2 mice as a short-term carcinogenicity study model and common marmosets as a small non-human primate species. We offer the optimal test systems in response to requests from clients such as using a combination of MRI and bioimaging.
【Activities】
- Administration of consigned drug efficacy and safety studies in NOG mice and rats
- Administration of consigned drug efficacy screening tests in the tumor-bearing mouse model
(Stock human tumor lines are also available.)
- Administration of consigned tumorigenicity studies of iPS cells, etc., in NOG mice
- Administration of therapeutic trial in the spinal cord injury model and the intracerebral administration study (Using common marmosets and/or nude rats)
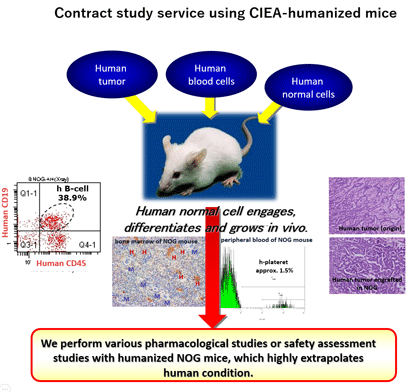
- Administration of various consigned studies using "humanized mice*".
- Distribution of uniquely-established and maintained human tumor lines (approximately 230 strains)
*Immunodeficient animals such as NOG mice with engrafted human cells
Contact
※References
-
Innate Response to Human
Cancer Cells with or without IL-2 Receptor Common γ-Chain Function in NOD Background Mice Lacking
Adaptive Immunity
Nishime C, Kawai K, Yamamoto T, Katano I, Monnai M, Goda N, Mizushima T, Suemizu H, Nakamura M, Murata M,
Suematsu M, Wakui M.
J Immunol. 2015 Aug 15;195(4):1883-90.
-
Characterization of in vivo
tumorigenicity tests using severe immunedeficient NOD/Shi-scid IL2Rγnull mice for detection of
tumorigenic cellular impurities in human cell-processed therapeutic products.
Kusakawa S, Machida K, Yasuda S, Takada N, Kuroda T, Sawada R, Okura H, Tsutsumi H, Kawamata S, Sato
Y.
Regen Ther. 2015 Feb 19;1:30-37.
-
Establishing a laboratory
animal model from a transgenic animal: rasH2 mice as a model for carcinogenicity studies in regulatory
science.
Urano K, Tamaoki N, Nomura T.
Vet Pathol. 2012 Jan;49(1):16-23.
-
In vivo chemotherapeutic
profile of human gallbladder small cell carcinoma.
Nishime C, Ohnishi Y, Suemizu H, Tamaoki N, Kusumi T, Sato F, Yamazaki H, Nakamura M, Ueyama Y, Kijima
H.
Biomed Res. 2008 Oct;29(5):251-6.
-
Gallbladder small cell
carcinoma Xenograft established by serial transplantation in nude mice.
Nishime C, Ohnishi Y, Suemizu H, Tamaoki N, Suematsu M, Oida Y, Yamazaki H, Nakamura M, Ueyama Y, Kijima
H.
Anticancer Res. 2006 Jan-Feb;26(1A):79-83.